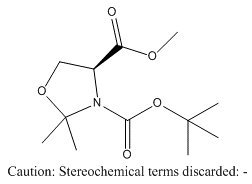
Methyl (S)-(-)-3-Tert-Butoxycarbonyl-2,2-Dimethyl-4-Oxazolidinecarboxylate
(S)-(-)-3-Tert-Butoxycarbonyl-4-Methoxycarbonyl-2,2-Dimethyl-1,3-Oxazolidine
CAS: 108149-60-6
Molecular Formula: C12H21NO5
Methyl (S)-(-)-3-Tert-Butoxycarbonyl-2,2-Dimethyl-4-Oxazolidinecarboxylate - Names and Identifiers
Methyl (S)-(-)-3-Tert-Butoxycarbonyl-2,2-Dimethyl-4-Oxazolidinecarboxylate - Physico-chemical Properties
| Molecular Formula | C12H21NO5 |
| Molar Mass | 259.3 |
| Density | 1.082g/mLat 25°C(lit.) |
| Boling Point | 101-102°C2mm Hg(lit.) |
| Flash Point | 199°F |
| Vapor Presure | 0.000629mmHg at 25°C |
| Appearance | Liquid |
| Color | Clear yellow |
| pKa | -3.58±0.60(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.443(lit.) |
| MDL | MFCD00192279 |
| Physical and Chemical Properties | WGK Germany:3 |
| Use | This product is for scientific research only and shall not be used for other purposes. |
Methyl (S)-(-)-3-Tert-Butoxycarbonyl-2,2-Dimethyl-4-Oxazolidinecarboxylate - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S37/39 - Wear suitable gloves and eye/face protection |
| UN IDs | NA 1993 / PGIII |
| WGK Germany | 3 |
| HS Code | 29349990 |
Methyl (S)-(-)-3-Tert-Butoxycarbonyl-2,2-Dimethyl-4-Oxazolidinecarboxylate - Reference Information
| Use | (S)-(-)-3-boc-4-methoxycarbonyl-2, 2-dimethyl-1, 3-oxazolidine is a useful intermediate for improving the synthesis of Ganal and its analogs, with a variety of pharmaceutical applications. At present, organic compounds containing oxazole ring as drugs have been widely used in clinical, and play an irreplaceable role in overcoming clinical drug resistance and treating infectious diseases. |
| preparation | the target compound (S) was prepared by one-pot reaction with ethanolamine, acetone and di-tert-butyl dicarbonate as starting materials.-(-)-3-boc-4-methoxycarbonyl-2, 2-dimethyl-1, 3-oxazolidine. The synthesis reaction scheme is as shown below: experimental procedure a thermometer, a dropping funnel and a condenser are respectively set on a 100ml dry three-diameter round-bottom flask, and ethanolamine, acetone and benzene are sequentially added thereto, followed by stirring at 35 ° C. For 30 minutes.. The reaction system is cooled to -10 ℃ with ice salt bath, and sodium hydroxide aqueous solution is added and stirred. When the reaction temperature drops to -5 ℃, di-tert-butyl dicarbonate is slowly added dropwise, and the temperature of the system is controlled to 0 ℃, the dropwise addition was completed in about 40 minutes, and stirring was continued for 1 hour. The reaction solution was washed with water until neutral, dried over anhydrous magnesium sulfate, the solvent was distilled off, and the solid was recrystallized from ethyl acetate/petroleum ether to obtain product (S)-(-)-3-boc-4-methoxycarbonyl-2, 2-dimethyl-1, 3-oxazolidine. |
Last Update:2024-04-09 21:22:08
Supplier List
Spot supply
Product Name: Methyl (S)-(-)-3-Boc-2,2-dimethyl-4-oxazolidinecarboxylate Visit Supplier Webpage Request for quotationCAS: 108149-60-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-(benzoylthio)-2-methylpropionic acid Request for quotation
CAS: 108149-60-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 108149-60-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (S)-(-)-3-TERT-BUTOXYCARBONYL-4-METHOXYCARBONYL-2,2-DIMETHYL-1,3-OXAZOLIDINE Request for quotation
CAS: 108149-60-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 108149-60-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: (S)-(-)-3-Boc-4-Methoxycarbonyl-2,2-Dimethyl-1,3-Oxazolidine Visit Supplier Webpage Request for quotation
CAS: 108149-60-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 108149-60-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Methyl (S)-(-)-3-Boc-2,2-dimethyl-4-oxazolidinecarboxylate Visit Supplier Webpage Request for quotationCAS: 108149-60-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-(benzoylthio)-2-methylpropionic acid Request for quotation
CAS: 108149-60-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 108149-60-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (S)-(-)-3-TERT-BUTOXYCARBONYL-4-METHOXYCARBONYL-2,2-DIMETHYL-1,3-OXAZOLIDINE Request for quotation
CAS: 108149-60-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 108149-60-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: (S)-(-)-3-Boc-4-Methoxycarbonyl-2,2-Dimethyl-1,3-Oxazolidine Visit Supplier Webpage Request for quotation
CAS: 108149-60-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 108149-60-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



